Meriscreen HIV 1-2 WB test kit
THIS IS A PROFESSIONAL KIT. FOR PURCHASE AND PRICING, PLEASE CONTACT US DIRECTLY.
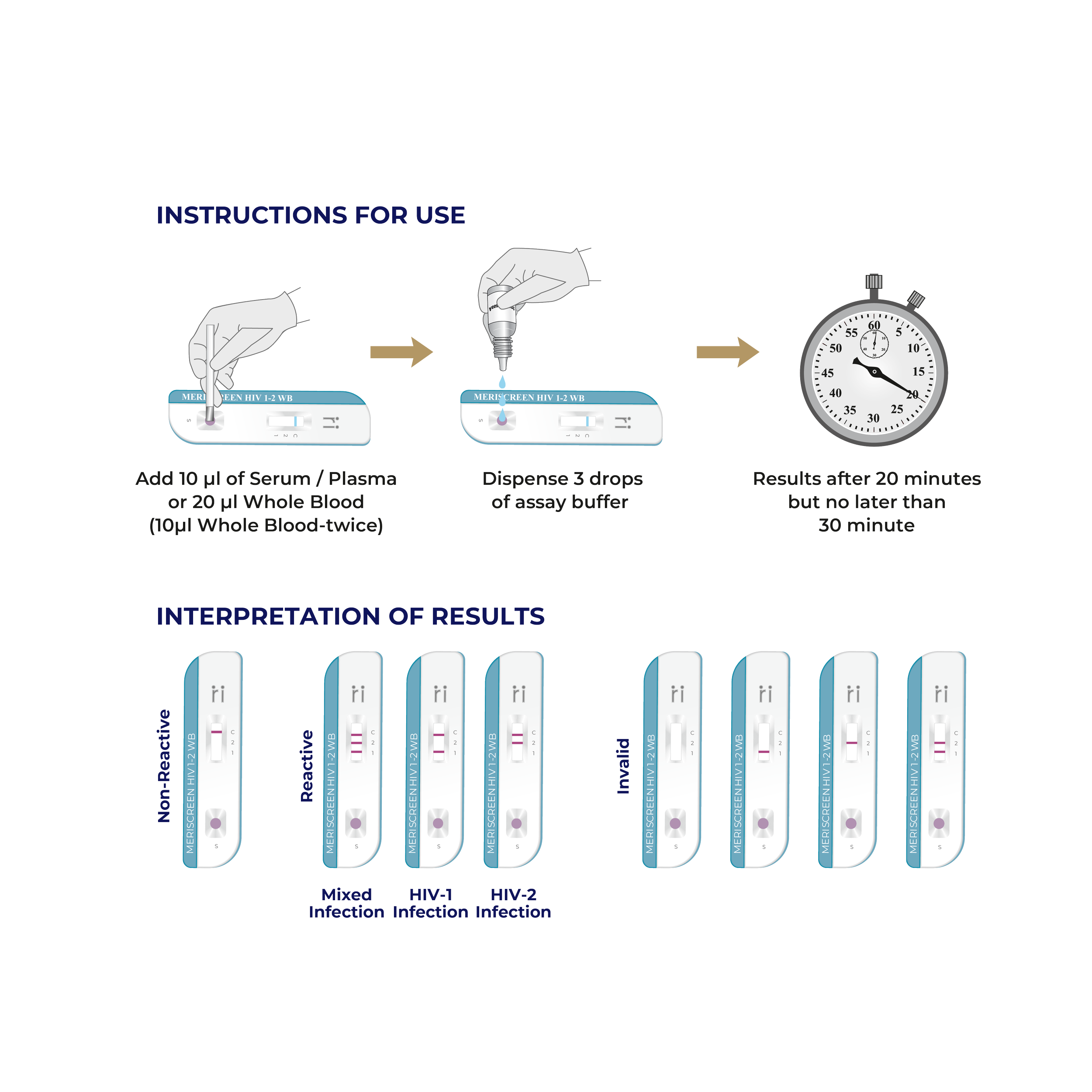
MERISCREEN HIV 1-2 WB Test is a single use, qualitative, screening, in-vitro diagnostic immunochromatography assay and used for detection of antibodies (IgG, IgA and IgM) specific to HIV-1 and HIV-2 in human fingerstick whole blood, venous whole blood, serum or plasma specimens.
MDA approved IVDD6688522-109236
ISO 13485:2016 QMS certification
CE marking
WHO prequalified IVD
Made in India
BENEFITS
High Accuracy
MERISCREEN HIV 1-2 WB Test has an impressive diagnostic sensitivity and specificity of 100.00%. (1)
Easy, Clear, Rapid Result
MERISCREEN HIV 1-2 WB Test provide results quickly, usually within 20 to 30 minutes. This enables immediate diagnosis and counselling.
WHO Prequalified in vitro diagnostics (IVD) Product for HIV Testing
MERISCREEN HIV 1-2 WB Test is WHO prequalified. It meets rigorous quality and performance standards, reducing the risk of inaccurate test results and ensures the reliability of HIV diagnosis.
Flexible
MERISCREEN HIV 1-2 WB Test can be used with serum, plasma, or whole blood.
PERFORMANCE
▲ MERISCREEN HIV 1-2 WB Test is a 3rd generation antibody tests with estimated median window period of 22 days where 13-46% chance of false negative result may occur. By 90 days, it able to detect more than 99% of the antibodies with 0 -1% chance of false negative result.(2)
SPECIFICATIONS
CONTENTS
30 Tests/Box (HWRPD-01)
30 Test device, pouched with desiccant
2 Assay buffer bottles (3.0mL)
30 Specimen transfer device – capillary tubes (10µL)
30 Sterile lancets
30 Alcohol swabs
1 Package insert
40 Tests/Box (HWRPD-02)
40 Test device, pouched with desiccant
2 Assay buffer bottles (3.0mL)
40 Specimen transfer device – capillary tubes (10µL)
40 Sterile lancets
40 Alcohol swabs
1 Package insert
REFERENCES
1. Meril Diagnostics Pvt., Ltd. (2020). Instructions for Use: MERISCREEN HIV 1-2 WB Test Package Insert.
2. Taylor D, Durigon M, Davis H, et al. Probability of a false-negative HIV antibody test result during the window period: a tool for pre- and post-test counselling. Int J STD AIDS. 2015;26(4):215-224. doi:10.1177/0956462414542987.